Introduction: The role of complement is increasingly recognized in disease pathogenesis, however diagnostic assays are limited by low predictive value, technical requirements, and inability to assess relative contributions of each pathway of complement (classical, alternative, or lectin). The traditional mHam was developed as a functional assay of complement-mediated eukaryotic cell death in atypical hemolytic uremic syndrome (aHUS). However, broader use of the assay has been limited by inter-operator dependence and technical requirements. We have developed a bioluminescent mHam that is robust and easily performed (no cell washes or exogenous reagent addition) and utilizes an autonomously bioluminescent HEK293 cell line to allow for real-time assessment of changes in metabolic health due to complement activation. Given both HEK293 and endothelial cells lack complement receptor 1 (CR1), a crucial regulator of the classical pathway (CP), this assay measures complement activity of greater relevance to disease pathogenesis.
Methods: The bioluminescent mHam uses LiveLight (LL) HEK293 cells (490 BioTech) engineered to stably express the bacterial luciferase ( lux) and flavin reductase genes ( luxF) to create autonomous bioluminescence. A PIGA null clone (LL PIGA- HEK293) was created through proaerolysin selection (removing GPI-anchored complement regulators CD55 and CD59). To perform the assay, 40,000 cells were harvested with trypsin/EDTA and plated as triplicates in gelatin veronal buffer (GVB ++,80 µL). Serum (20 µL) was added after specified treatments (heat inactivation or inhibitor addition). Activity was monitored by serial luminescence measurements every 5 min at 37 °C. A relative luminescence of <25% at one hour is designated as positive based upon lowest replicate of any healthy control.
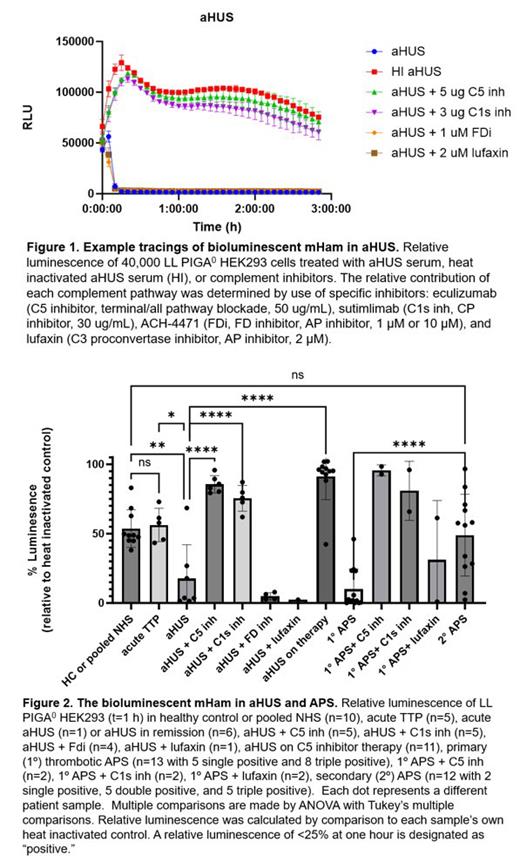
Results: The bioluminescent mHam, unlike the original mHam, provides continuous measurement of complement activity (example tracing Figure 1). The complement specificity of the luminescent changes were demonstrated with heat inactivation, terminal complement blockade with eculizumab, and calcium/magnesium dependence. The relative contribution of CP to changes in luminescence was confirmed with the specific CP inhibitor sutimlimab as well as use of pathway specific buffers. Healthy control (HC) samples show a relative luminescence of 53.7% (95%CI 44.0-63.4) with no difference observed in acute thrombotic thrombocytopenic purpura (TTP) (mean 56.2%, 95%CI 41.2-71.2, p=>0.9999). We next utilized samples of aHUS patients known to be in remission, off complement inhibitor therapy. A large proportion of these patients have ongoing assay positivity even in the traditional mHam (9 of 13 in one unpublished cohort). The bioluminescent mHam shows that 5 of 6 aHUS patients in remission have ongoing positivity (mean 17.56%, 95%CI -10.5-45.6, p=0.0131). An acute aHUS sample showed similar results (17.8% after plasmapheresis). Unexpectedly, the relative luminescence is rescued by addition of either C1s or C5 inhibitors, but use of alternative pathway (AP) inhibitors has no significant rescuing effect ( Figure 1-2). Of aHUS patients tested while on complement inhibition, 91% (10/11) show greater than 88% blockade ( Figure 2 aHUS on therapy) . Finally, in one cohort, the traditional mHam was positive in 52% (14/27) of primary antiphospholipid syndrome (PAPS) cases and 15% (10/66) of secondary APS (SAPS) (unpublished data). Thus far, the bioluminescent mHam shows abnormal complement activity in 92% (12/13) of PAPS and 17% (2/12) of patients in SAPS ( Figure 2). Again, complement inhibition with C1s or C5 inhibitors abrogated the effect in a dose dependent manner.
Conclusions: The bioluminescent mHam test is an exquisitely sensitive real-time measure of abnormal complement activity. It is easy to perform, more closely approximates the complement regulators on renal endothelium, can be utilized for therapeutic monitoring, and has already offered novel disease insights through the use of specific pathway inhibitors. Although aHUS is traditionally categorized as a disorder of the AP, these results suggest the “trigger” mediating disease pathology is the classical pathway. Also, we show that a large proportion of primary APS patients have abnormal CP activation responsive to C1s and C5 inhibition, suggesting the disease may be responsive to complement-based therapeutics.
Disclosures
Gerber:Merck: Honoraria; Pfizer (spouse): Current Employment, Current equity holder in publicly-traded company; Alexion Pharmaceuticals: Consultancy; Apellis Pharmaceuticals: Consultancy. Chaturvedi:Alexion: Consultancy, Other: Advisory board participation; Sobi: Honoraria; Takeda: Other: Advisory board participation; Sanofi Genzyme: Consultancy; Sanofi: Other: Advisory board participation. Brodsky:Alexion, AstraZeneca Rare Disease: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal